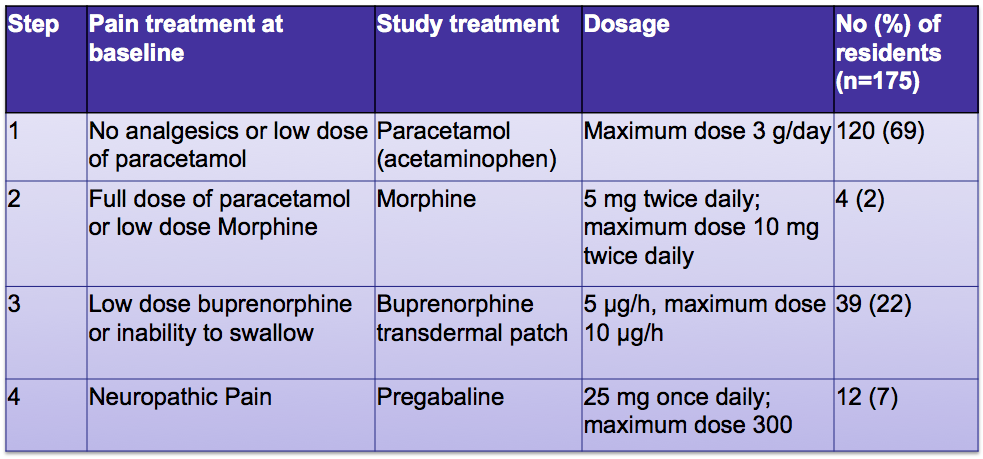
SUMMARY OF PRODUCT CHARACTERISTICS 1. NAME OF THE MEDICINAL PRODUCT <invented name> 4.6 mg/24 h transdermal patch <inve

PDF) Safety and tolerability of transdermal and oral rivastigmine in Alzheimer's disease and Parkinson's disease dementia

PDF) A pilot randomized controlled trial evaluating an integrated treatment of rivastigmine transdermal patch and cognitive stimulation in patients with Alzheimer's disease | Daniele Sancarlo and Francesco Panza - Academia.edu